Adding Tasks
Tasks can be created by clicking on one of many links. The Club/Tasks/Create a task menu item is one way. This will initialize a task with you assigned to the task. You can change this by removing yourself or adding others to the assigned list.
The other way to create a task is to click on the Tasks tab when viewing a node. You then see the task list associated with the node as well as a set of links (Fig. 1).
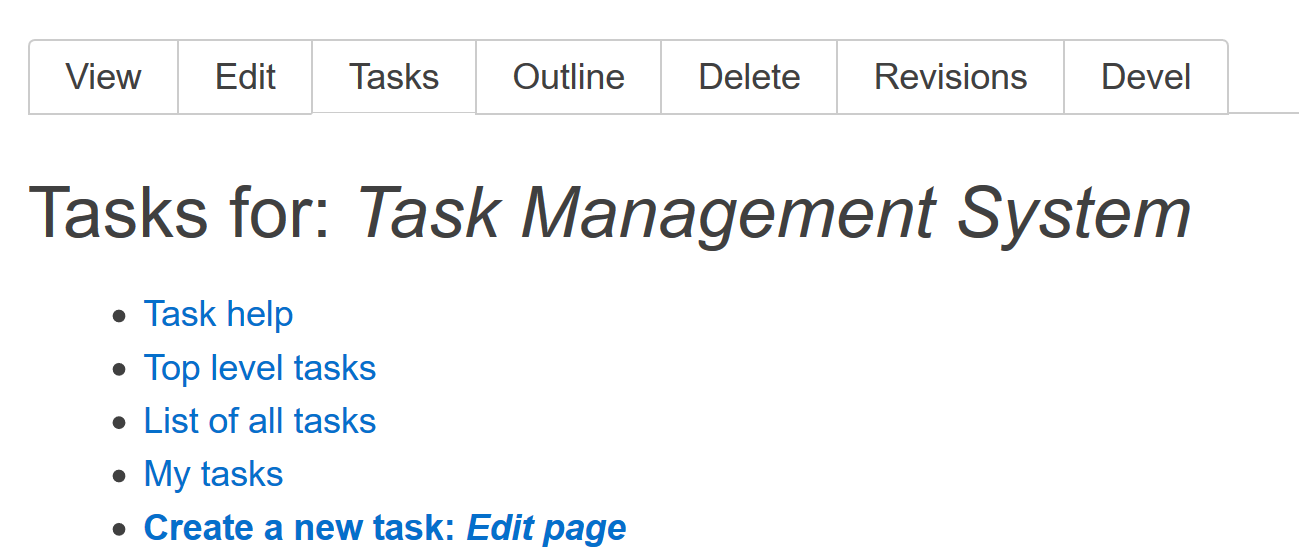
The Create a new task;: Edit page will create a task with the node title in it but you will have to add the node to the Related Content section if you want a reference to it. You can change anything when editing the task before you save it. There is also a create task link on the task lists that are not associated with a node. This includes Top level tasks and List of all tasks.
