The Effect of Water Temperature on Sugar Cube Decomposition Rate
Abstract:
Bibliography/Citations:
No additional citationsAdditional Project Information
Project files
Research Plan:
1.) Measure out 1 1/2 cups of tap water, and pour it into a microwave-safe clear container, use the thermometer to make sure the water is 52-55°F. If it is not, either heat the water with a microwave or make it colder with a fridge or freezer. (Be careful when dealing with hot water).
2.) Take a sugar cube and stop it into the container, when it hits the water start a stopwatch and stop it when the sugar cube fully decomposes. Write down your results in the data table. (Be sure to empty the water after every trial).
3.) Measure out 1 1/2 cups of tap water and check the temperature, if the water is too cold, heat the water to 77-80°F by using a microwave. Then pour the water into the clear container.
4.) Take a sugar cube and drop it into the container, as it hits the water start a stopwatch and stop it when the cube fully decomposes. Write down your results in the data table.
5.) Repeat steps 3-4, increasing the temperature by about 25*F for each trial.
6.) Optional: Take 2 clear containers at different temperatures of water, the coldest temperature of the trials, and the hottest. Drop a sugar cube in each clear container at the same time. Record a video of the sugar cube dissolving and observe the reactions.
Questions and Answers
1. What was the major objective of your project and what was your plan to achieve it?
a. Was that goal the result of any specific situation, experience, or problem you encountered?
b. Were you trying to solve a problem, answer a question, or test a hypothesis?
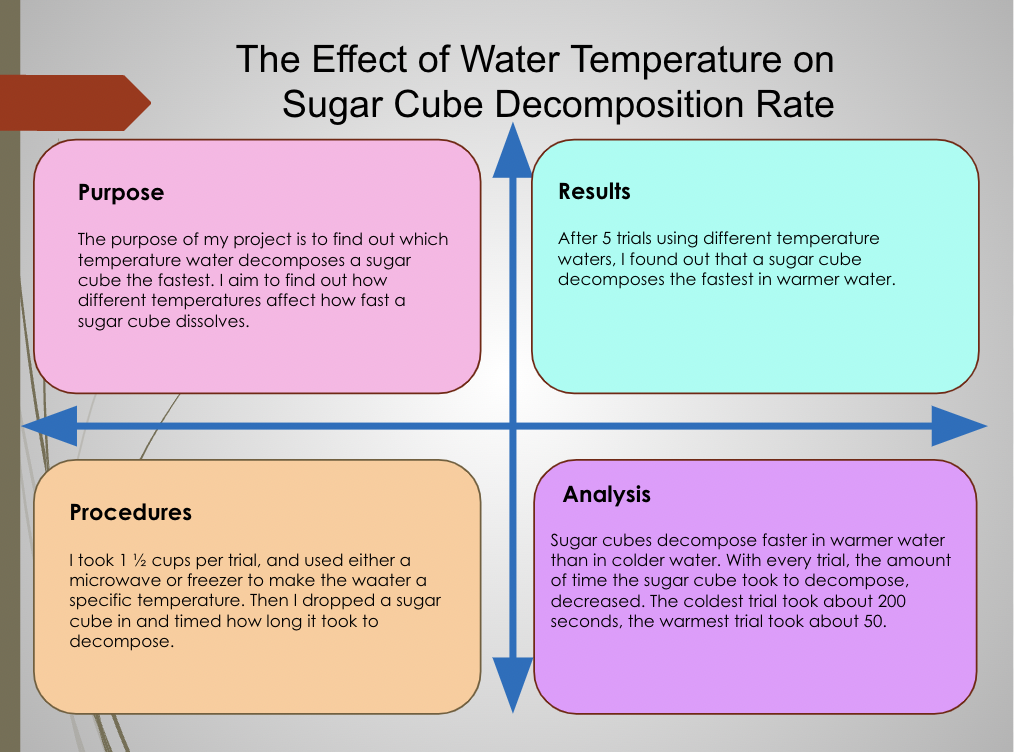
The major objective of my project was to discover which temperature of water best dissolves a sugar cube. My plan to achieve this was to fill a microwave-safe clear container with different temperatures of water for each trial. During each trial, I would drop a sugar cube in the container and start a stopwatch to time how long it would take for the sugar cube to decompose. I was trying to test a hypothesis. I predicted that the sugar cube would decompose faster in warmer water than in colder water.
2. What were the major tasks you had to perform in order to complete your project?
a. For teams, describe what each member worked on.
The major tasks I had to perform in order to complete my project were to drop the sugar cube in the water and time how long it took the sugar cube to decompose and heat the water to up to 150°F.
3. What is new or novel about your project?
a. Is there some aspect of your project's objective, or how you achieved it that you haven't done before?
b. Is your project's objective, or the way you implemented it, different from anything you have seen?
c. If you believe your work to be unique in some way, what research have you done to confirm that it is?
In the past, I have not experimented with reaction times, so it was slightly difficult to figure out how to create a procedure for this project. My procedure was similar to experiments I have conducted in the past because I included repetition and made it easy to understand the procedure. This experiment has been conducted in the past by others, but I wanted to try it myself to see if I got similar results.
4. What was the most challenging part of completing your project?
a. What problems did you encounter, and how did you overcome them?
b. What did you learn from overcoming these problems?
The most challenging part of completing this project was heating up and cooling down the water to a specific temperature. I overcame this problem by learning how long to heat it up in the microwave and learning how long to keep it in a fridge or freezer. I learned that it is tricky to use water and temperature in an experiment, but it gives accurate results.
5. If you were going to do this project again, are there any things you would do differently the next time?
I could improve the method by adding another person to help in some parts of the procedure. I would make this improvement because it was a bit of a struggle with starting the stopwatch and dropping in the sugar cube at the same time, though it could have something to do with the type of stopwatch I used. I would also conduct another trial, with colder water than 52-55 degrees Fahrenheit to get more accurate results.
6. Did working on this project give you any ideas for other projects?
Yes, this project gave me another idea for other projects. An experiment that I could conduct in the future would be testing to see if sugar cubes decompose faster in different types of water if there is any variation in the results. I also want to experiment with reaction times, it may be with materials or people by using a stimulus and response.
7. How did COVID-19 affect the completion of your project?
COVID-19 did not affect the completion of my project because my experiment was simple and used household materials.