Shape-tunable Gold Nanoparticles for Combined Circulating Tumor DNA Capture and Plasmonic Detection
Abstract:
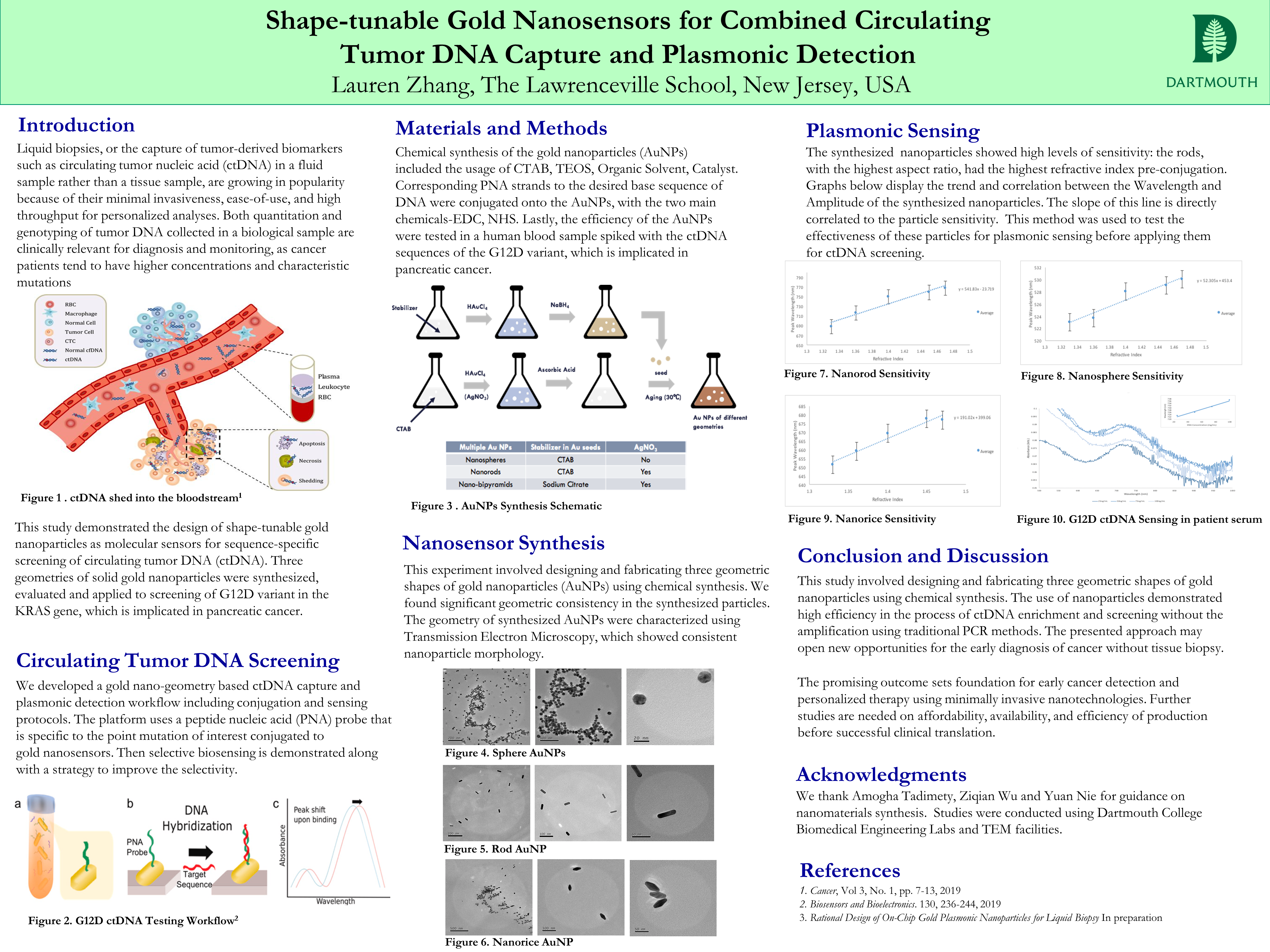
This study involved the development of gold nanoparticles with various geometries conjugated with Peptide Nucleic Acid (PNA) probes for fast enrichment of circulating tumor DNA (ctDNA) and plasmonic readout. ctDNA is gaining popularity as a potential biomarker for liquid biopsy, the process of detecting molecules in the peripheral blood rather than a tissue sample. It has wide ranging applications as a diagnostic and prognostic biomarker with a similar mutational profile as the tumor. However, it is challenging to detect ctDNA due to its small size and low concentration. In addition, the point mutations under investigation are typically difficult to be differentiated from wild type sequences. This study involved designing nanoscale sensors with combined molecular recognition and optical readout functions to address above challenges. Specifically, we design and fabricate three geometric shapes of gold nanoparticles using chemical synthesis. These nanosensors are then functionalized on the surface to identify biomarkers of interest. The gold-air interface also offers unique plasmonic properties which are sensitive to the local presence of the sequence-specific ctDNA binding. We demonstrate sequence-selective ctDNA screening through screening clinically relevant variants in the KRAS gene. The outcome of this project demonstrates novel designs of nanosensors with high efficiency in the process of ctDNA enrichment and screening without the amplification using traditional PCR methods. The presented approach may open new opportunities for the early diagnosis and personalized therapy of Cancer without invasive tissue biopsy.Bibliography/Citations:
No additional citationsAdditional Project Information
Research Plan:
In this study, we work to synthesize multiple shapes of gold nanoparticles and apply them to sensing clinically relevant sequences of ctDNA. Wee first work on a synthesis workflow and characterization using electron microscopy. Then we evaluate the resonance spectra through spectrometer measurements of light absorbance through the nanoparticles. We characterize the absorbance through the particles in varying concentrations of water and glycerol to understand how sensitive the particles are to changes in refractive index. We then apply this system to conjugation of the particles in solution, and testing with increasing concentrations of synthetic ctDNA in patient serum samples.
Phase I:
Task 1: Design and synthesize novel plasmonic nanoparticles through shape-controlled synthesis
Task 2: Initial development of numerical simulation to calculate plasmonic enhancement of novel nanoparticle geometries
Deliverable 1: SEM/TEM images of fabricated nanoparticles and their optical resonance spectra, with preliminary simulation data.
Phase II
Task 1: Determine bulk refractive index sensitivity of newly fabricated nanoparticles and compare to electromagnetic simulation
Task 2: Develop conjugation chemistry for PNA attachment to plasmonic nanoparticles
Task 3: Initial development of microfluidic array formation of novel nanoparticles
Deliverable 1: Bulk refractive index sensitivity of novel nanoparticles and comparison to simulation, with conjugation protocols and calibration curves.
Deliverable 2: SEM/TEM images of arrayed nanoparticles and microfluidic channels used for array formation.
Phase III
Task 1: Develop calibration curve for KRAS mutation sensing in buffer with array type nanoparticle structures to determine plasmonic sensitivity
Task 2: Test array structures with spiked patient serum samples
Task 3: Begin to multiplex to capture 3-5 mutations at once on a single chip
Deliverable 1: Calibration curves of three nanoparticle array type structures and relative sensitivities with comparison to simulation.